
Drinking and sniffing rubbing alcohol for its intoxicating properties is a trend.
Commercial rubbing alcohol preparations made from isopropyl alcohol (isopropanol) or ethanol are cheaper and easier to obtain. Thus, people unable to get alcoholic beverages–such as young people under the legal age limits–may resort to rubbing alcohol preparations to get drunk.
Adults who can obtain alcoholic beverages may also drink or sniff isopropyl alcohol-based rubbing alcohol as a more potent substitute for conventional alcoholic beverages.
This article offers detailed answers to your questions about the dangers of drinking or inhaling rubbing alcohol. We also list the signs and symptoms of poisoning.
Contents:
Rubbing alcohol is also made from denatured alcohol
Why do some people drink rubbing alcohol?
What happens when you drink rubbing alcohol?
Why do some people sniff rubbing alcohol?
What happens when you sniff rubbing alcohol?
Sniffing rubbing alcohol and anemia
Sniffing rubbing alcohol for nausea
Signs and symptoms of poisoning
–Gastrointestinal signs and symptoms
–Cardiovascular and respiratory system symptoms
Isopropanol toxicity compared with ethanol and methanol
Why ethanol-based rubbing alcohol could be dangerous
Comparative toxicity of isopropyl alcohol, ethanol, and methanol
What is rubbing alcohol?

Rubbing alcohol is typically a mixture of isopropyl alcohol and water used primarily for its antiseptic and disinfectant properties.
Isopropyl alcohol (ISO) (also known as isopropanol or 2-propanol) is a colorless, bitter-tasting, and toxic liquid with antibacterial properties.
The chemical formula is C3H8O (CH3CHOHCH3 or (CH3)2CHOH).
Most commercial preparations of rubbing alcohol contain a 70% aqueous solution of isopropyl alcohol.
Rubbing alcohol is a common household topical (skin) antiseptic for cleaning superficial bruises and cuts as part of first aid treatment to prevent infection.
Household use also includes skin sanitization and disinfection of household objects and surfaces people touch frequently. That includes kitchen and washroom countertops, light switches, doorknobs, and laptop keyboards.
Some brands of rubbing alcohol contain ethanol (ethyl alcohol; C2H6O) instead of isopropyl alcohol. Ethanol is not the same substance as isopropyl alcohol. It is the form of alcohol found in alcoholic beverages, such as beer, wine, and liquor (distilled spirits).
Rubbing alcohol containing isopropyl alcohol or ethyl alcohol is generally used for the same purposes in household and industrial settings.
The term “rubbing alcohol” is most commonly used in the U.S.
In the U.K., it is better known as “surgical spirit.” Surgical spirit is also a household sanitizer, antiseptic, and disinfectant.
Active ingredients
Rubbing alcohol for household use typically contains about 70% of isopropyl alcohol (isopropanol, C3H8O.). The balance of 30% consists of water and other ingredients, such as stabilizers and color additives.
Some brands may have isopropyl alcohol concentrations significantly higher than 70%, while others may have as low as 50%. Others contain methyl salicylate, an essential oil known as “wintergreen oil.”
Rubbing alcohol containing methyl salicylate (“wintergreen oil”) is a liniment for relieving muscle and joint aches and pain.
Rubbing alcohol is also made from denatured alcohol

Denatured alcohol is ethanol (ethyl alcohol) mixed with toxic, unpleasant-tasting (bitterants), or foul-smelling additives to discourage consumption.
Denaturing additives include methanol (methyl alcohol), benzene, pyridine, denatonium (a bitterant), castor oil, gasoline, and acetone.
[Fun fact: Denatonium salts– available as benzoate and saccharide– are the most bitter substances known.]
Methanol is very toxic to humans. It is the most commonly used additive for denaturing ethanol to prevent consumption. That explains why denatured alcohol products are known as “methylated spirits” in Canada, Britain, and Australia.
Preparations containing 100% isopropyl alcohol (ISO) aren’t suitable for household use as rubbing alcohol.
ISO is a skin irritant. It is harsher on the skin in high concentrations. Concentrated solutions are also more highly flammable and toxic than diluted forms.
You are more likely to suffer toxic effects from skin or eye contact, ingesting, and inhaling 100% ISO.
However, 100% isopropanol is the starting material for manufacturing rubbing alcohol, cosmetics, pharmaceuticals, cleaners, dye solutions, antifreeze products, soaps, inks, and window pane cleaners.
Why do some people drink rubbing alcohol?

Rubbing alcohol containing isopropanol is among the most common causes of alcohol poisoning in the United States.
Most cases involve children younger than 6 years who unintentionally (Slaughter et al., 2014) ingest the clear liquid after mistaking it for drinking water.
However, cases involving adults intentionally ingesting it are also common.
Ashurst and Nappe (2022) reported that about 80% of cases reported to the American Association of Poison Control Centers were unintentional.
Isopropyl alcohol has intoxicating properties similar to ethanol.
Ethanol is the intoxicating agent in commercially alcoholic beverages. However, isopropyl alcohol is a more potent intoxicant than ethanol. Thus, some people — especially people struggling with alcohol addiction— may take it as a cheaper and more available alternative.
Anyone can purchase a bottle of rubbing alcohol from a store or pharmacy at a lower cost than the same volume of conventional alcoholic beverages.
Alcohol is a highly addictive drug. People struggling with addiction and who have developed tolerance to the intoxicating effects of ethanol may turn to isopropanol to experience a more potent intoxicant.
It is absorbed into the bloodstream more rapidly than ethanol. Thus, it induces intoxication faster and more intensely.
Others may ingest it to commit self-harm.
Some cases are associated with pathological cravings, such as pica (allotriophagia).
Sharma and Morrow (2017) reported a case of a 36-year-old woman diagnosed with severe substance abuse involving the compulsive consumption of rubbing alcohol swabs.
Pica may also be associated with iron deficiency anemia.
What happens when you drink rubbing alcohol?
Rubbing alcohol is absorbed into the system through the oral route, inhalation, and skin contact.
We shall first discuss the metabolism and health effects of drinking rubbing alcohol.
You may scroll down here to read about how the metabolism of inhaled rubbing alcohol differs from when taken through the mouth.
We shall also later discuss the health effects of inhaling or sniffing it.
Why do some people sniff rubbing alcohol?
People inhale rubbing alcohol preparations containing both ethanol and isopropyl alcohol.
We focus primarily on the effects of sniffing rubbing alcohol preparation containing isopropyl alcohol. However, the effects of inhaling ethanol-based preparations are comparable to sniffing ISO-based products.
People sniff rubbing alcohol for much the same reasons as they drink it.
Alcohol is a highly addictive drug. It ranks alongside drugs, such as nicotine (freebase or salts) and weed (THC containing C. sativa), as one of the most widely abused psychoactive drugs. Thus, the habit of sniffing rubbing alcohol could quickly become an addiction.
Experts consider alcohol, nicotine and marijuana as gateway drugs. They boost dopamine levels in susceptible individuals. Increased tolerance to the effects of gateway drugs leads users to see new psychoactive substances.
People struggling with alcohol use disorder or addiction may prefer sniffing isopropyl alcohol-based preparations due to their more potent intoxicating effect.
What happens when you sniff rubbing alcohol?
Inhaling alcohol (either as isopropyl alcohol or ethanol) became a trend among college students and other consumers of alcohol some years back.
A method involved heating alcohol or pouring it over dry ice and inhaling the vapors to induce rapid and intense intoxication.
Some soak cotton balls or swabs in an alcohol solution and sniff it. Others vape it.
We previously discussed how alcohol ingested through the oral route gets metabolized (broken down) in the liver and rapidly eliminated from the system through the kidneys, thus reducing the risk of deadly poisoning.
The digestive system also has mechanisms, such as vomiting, that protect the body against a lethal load of alcohol when ingested through the oral route.
However, inhaled alcohol passes via the lungs directly into circulation and the brain, sidestepping the digestive system and the liver which help to protect against poisoning.
Inhaling is thus more dangerous than drinking because it increases the risk of fatal poisoning.
Some people also reportedly sniff alcohol because they believe that bypassing the digestive system prevents added nutritional calories (alcohol metabolism in the liver releases carbon dioxide, water, and energy/calories). But experts have noted that this belief is mistaken because inhaled alcohol is eventually metabolized in the liver to release energy before excretion.
Inhaling can also cause brain damage and injure lung tissues.
Sniffing rubbing alcohol and anemia
Drinking or inhaling rubbing alcohol could be associated with psychopathological cravings, such as pica or obsessive-compulsive disorder.
Iron deficiency anemia is associated with strange cravings, including pica (learn more about strange cravings associated with anemia here).
People with iron deficiency anemia may also experience a compulsive desire to sniff or smell various substances.
A compulsive desire to sniff or smell volatile substances, such as alcohol, gasoline, and menthol, is known as desiderosmia (olfactory craving).
Sniffing rubbing alcohol for nausea and vomiting
Studies suggest that inhaling or sniffing isopropyl alcohol may improve mild-to-moderate nausea and vomiting among emergency department (ED) patients.
Beadle and associates (2015) reported that inhalation relieved nausea among ED patients, compared with placebo.
According to Lindblad and co-workers (2018), a trial involving nonpregnant women found that inhaling or smelling isopropyl alcohol significantly improved nausea scores.
Candemir and colleagues (2020) also found that isopropanol was more effective than a placebo for treating nausea and vomiting.
But despite these findings, you should never self-treat nausea at home by inhaling or sniffing isopropyl alcohol because it is dangerous. These studies were only trials investigating potential treatments. Doctors do not recommend inhaling rubbing alcohol at home to relieve nausea and vomiting.
Treating nausea at home by sniffing rubbing alcohol could cause severe damage to your health.
Consult your healthcare provider if you have persistent nausea and vomiting. They will investigate the cause of your symptoms and prescribe the best treatments.
Isopropyl alcohol metabolism
Rubbing alcohol is a more potent intoxicant than ethanol when taken by oral route. The CNS effects are partly to differences in metabolic pathways compared with ethanol.
Unlike ethanol, one of the end-products of the breakdown of isopropyl alcohol is acetone, a potent CNS depressant.
Isopropyl alcohol (ISO) is rapidly absorbed, with plasma concentration levels peaking between 30 minutes after ingestion (Slaughter et al., 2014).
When taken without ethanol, ISO has a half-life of 2.5-8 hours.
The liver metabolizes about 80% of the ingested substance (Ashurst and Nappe, 2022).
The enzyme alcohol dehydrogenase breaks ISO down in the liver to acetone.
However, taking it with ethanol prolongs the substance’s half-life because alcohol dehydrogenase metabolizes ethanol preferentially.
Acetone is a toxic by-product of alcohol dehydrogenase metabolism of isopropanol. It has a half-life of 7.7-27h (Slaughter et al., 2014) following ingestion of ISO.
Alcohol dehydrogenase metabolizes isopropanol to produce the following metabolites excreted through the kidneys:
- Acetone
- Acetol
- Methylglyoxal
- Propylene glycol
- Acetate
- Formate
The metabolites listed above may convert to glucose and other products.
Although most isoproproanol excretion is through urine, some occurs through the lungs (pulmonary excretion) alongside acetone.
Part of the absorbed isopropanol is eliminated unchanged in the urine (Slaughter and associates (2014). The balance gets converted to acetone and metabolites of acetone.
Acetone metabolism occurs in the liver and excretion through the lungs (breath) and urine (Umeh al., 2021).
Toxicity
The toxicity of rubbing alcohol is due primarily to the combined effect of isopropyl alcohol and its acetone metabolite (Slaughter et al., 214).
While the details of the mechanism of toxicity are not well understood, we know that ISO is a powerful CNS depressant and may induce sedation, coma, and death.
It is potentially lethal at doses of 2 to 4 ml/kg (Ashurst and Nappe, 2022).
The metabolic conversion to acetone (dimethyl ketone) enhances its CNS depressant activity.
Although ingesting isopropyl alcohol may cause death, lethal outcomes are rare compared with methanol (Ashurst and Nappe, 2022; Slaughter et al, 2014).
Most cases of poisoning occur in children younger than 6 years old.
Death due to poisoning is usually due to the toxic effects of the accumulation of the substance and its acetone metabolite in the system.
Signs and symptoms of poisoning

The signs and symptoms of rubbing alcohol poisoning via oral ingestion and sniffing/inhalation are similar to those of ethanol.
We list the symptoms of poisoning under three categories:
- Gastrointestinal (GI)
- Central Nervous System (CNS)
- Cardiovascular and respiratory
Gastrointestinal signs and symptoms
Isopropyl alcohol is a digestive system or gastrointestinal irritant. Digestive system symptoms of poisoning include:
- Abdominal pain
- Nausea and vomiting blood
- Hematemesis: Vomiting blood due to bleeding in the GI tract
- Hemorrhagic gastritis: Loss of stomach linings (mucosa) and bleeding. The effect may spread to the intestines.
- Diarrhea
- Rupture of the bladder
Poisoning may lead to organ damage (liver and kidney damage) and death.
CNS symptoms
Isopropanol and its metabolite acetone are potent central nervous system (CNS) depressants. Signs and symptoms associated with CNS toxicity include:
- Dizziness
- Headache
- Sedation
- Headache
- Inebriation or drunkenness (confusion, slurred speech, gait, and postural disturbances, such as staggering and falling over)
- Altered sensorium: Confusion, emotional, thought process, and behavioral disturbances, disorientation, poor judgment, memory loss, loss of alertness or consciousness,
- Hallucinations
- Brain and nerve damage
- Hearing loss
- Loss of corneal and deep tendon reflexes
- Loss of coordination and limb spasms
- Loss of consciousness
These signs and symptoms may lead to coma and death.
Cardiovascular and respiratory system symptoms
- Depressed cardiovascular function
- Hypotension may be severe and associated with increased mortality risk
- Internal hemorrhage
- Hemodynamic instability
- Respiratory arrest
- Cardiac arrest
The signs and symptoms may lead to shock and death.
Isopropanol toxicity compared with ethanol and methanol

To put the toxicity of isopropanol in perspective, we shall compare it with ethanol, the active alcohol in commercial alcoholic drinks (read: Comparative toxicity of isopropyl alcohol, ethanol, and methanol).
Ethanol is less toxic than isopropyl alcohol because alcohol dehydrogenase metabolizes it to form acetaldehyde, a compound that can be rapidly removed from the system (see schematic illustration of ethanol metabolism here).
Acetaldehyde is broken down and excreted through multiple oxidate steps that end with the release of carbon dioxide, water, and energy.
Ethanol metabolism fun facts
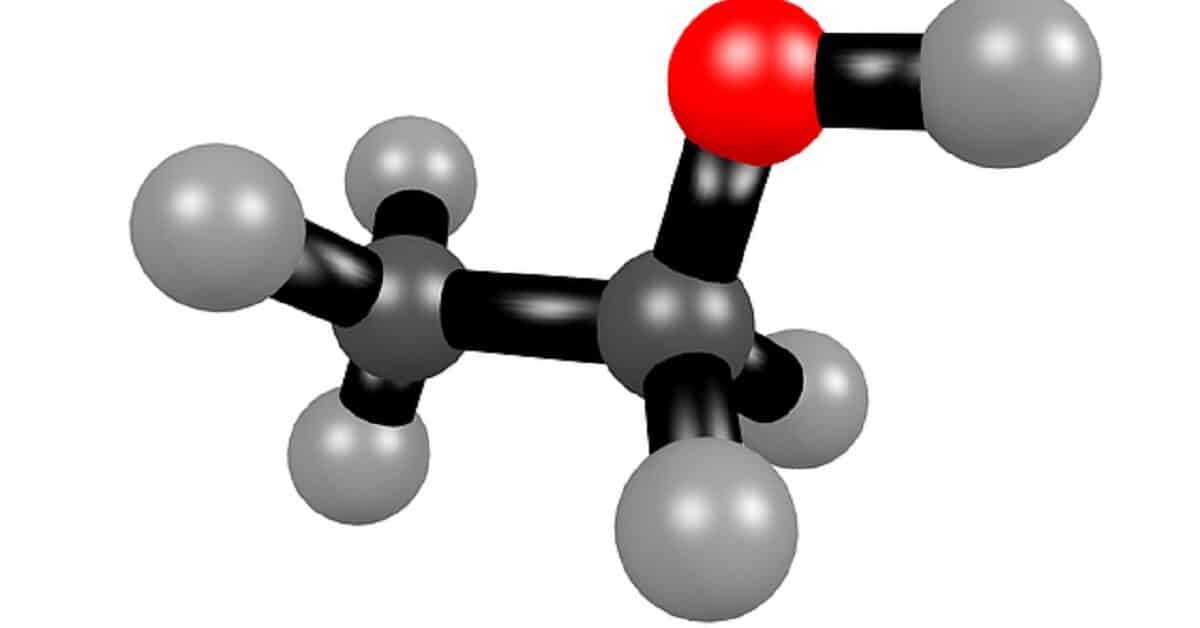
- In the liver, alcohol dehydrogenase (ADH) catalyzes the oxidative metabolism of ethanol to acetaldehyde
- Aldehyde dehydrogenase (ALDH) then catalyzes the oxidization of acetaldehyde to form acetate
- Acetate converts to Acetyl CoA. The conversion, which occurs in the mitochondria, is catalyzed by Acetyl-CoA synthetase 2, ACSS2
- Acetyl-CoA passes into the citric acid cycle. Metabolic breakdown of Acetyl-CoA in the mitochondria yields carbon dioxide, water, and energy released (compare glucose metabolism).
- The process is represented in a simplified form as shown below:
- C2H6O(ethanol) → C2H4O(acetaldehyde) → C2H4O2(acetate/acetic acid) → acetyl-CoA → 3H2O + 2CO2 (see schematic illustration here)
Why ethanol-based rubbing alcohol could be dangerous
Ethanol-based rubbing alcohols may contain methanol.
Methanol is used as a denaturant to discourage the consumption of ethanol-based products. Denatured alcohol (methylated spirit) may contain 5-10% methanol.
Methanol (methyl alcohol) is a highly toxic chemical. Handle rubbing alcohol containing the substance with care.
Methanol is more toxic than ethanol and isopropyl alcohol because alcohol dehydrogenase metabolizes it to form formaldehyde (Chan and Chan, 2018; Skrzydlewska, 2003).
Aldehyde dehydrogenase then metabolizes formaldehyde to form formic acid. Formic acid metabolism yields folic acid, folinic acid, carbon dioxide, and water.
These acid end-products are highly toxic to the human system. The accumulation of acids in the body may cause metabolic acidosis, leading to blindness and death.
Methanol (also known as wood alcohol) is a constituent of multiple commercial products. Varnishes, paints, washer fluids, antifreeze, and adhesives contain methanol.
Methanol is so poisonous that ingesting as little as 30 to 240 ml could be lethal. The median lethal dose is estimated to be 100ml or 1-2 ml/kg.
For comparison, isopropyl alcohol has a lethal dose range of 2-4 ml/kg. Experts have advised manufacturers not to add methanol to rubbing alcohol (Chan and Chan, 2018).
Keep ethanol denatured with methanol (methylated spirit) away from young children.
According to Chan and Chan (2018), repeated use of methylated spirit as a hand rub could result in chronic toxicity due to skin absorption.
The signs and symptoms of methanol toxicity include loss of vision, breathing difficulty, hypotension, seizures, and coma.
Comparative toxicity of isopropyl alcohol, ethanol, and methanol
The consumption of only 10 ml of pure methanol may result in permanent blindness, and as little as 30 ml may be lethal.
By comparison, 160-240 ml of isopropyl alcohol is potentially lethal, depending on alcohol tolerance and weight of the consumer.
Consumption of 0.5 to 1 ml/kg of 70% isopropyl alcohol (ISO) solution may only have non-lethal toxic effects.
Dosing of 2-4ml/kg pure ISO is potentially lethal, but adults may survive higher doses. [Note: 2-4ml/kg is equivalent to about 140-280 ml, assuming a body weight of about 70kg.]
In adults, moderate to severe symptoms of intoxication may occur after ingestion of 1 to 1.5 ml/kg of 95% to 99% ethanol.
Thus, depending on alcohol tolerance, about 70-100 ml of ethanol may cause moderate to severe intoxication in an adult with an average body weight of about 70kg.
Chronic ethanol consumers may require higher doses.
Ingestion of 2 to 3ml/kg (≈2-3g/kg or140-200ml) will cause severe symptoms of intoxication even in chronic consumers (assuming, once again, a body weight of about 70kg).
Death may occur in adults after ingesting 5-6ml/kg (350-400ml). But chronic consumers may survive and recover even after taking higher doses.
Poisoning treatment

Commercially available isopropanol- or ethanol-based rubbing alcohol products are sanitizers and disinfectants, not beverages.
Never drink or inhale them because they are toxic. They are not substitutes for liquor, wine, and other alcoholic beverages.
We have discussed in detail the toxicity of rubbing alcohol in previous sections of this article (read: Rubbing alcohol toxicity).
If you or someone you know has ingested or inhaled rubbing alcohol purposely or accidentally, they need prompt medical attention.
Call 911 immediately. Also, call Poison emergency number 1-800-222-1222 (U.S.) or PoisonHelp.org.
Experts will guide you on first aid you may administer to the victim before help arrives.
Canadian readers may view the poison help emergence numbers in their respective provinces here.
Poison control experts may ask for information about the victim, such as age, body weight, how much the victim swallowed, and when the incident happened.
Follow their instructions closely.
If the poison has been in contact with the victim’s eyes or skin, wash the affected area with water for up to 15 minutes.
Don’t induce vomiting because it may cause complications.
To prevent poisoning at home, keep all rubbing alcohol products out of the reach of children.
If you or someone you live with is struggling with alcohol addiction, it is best to keep your home alcohol-free. That may also include ensuring they can’t get access to products such as rubbing alcohol.
People struggling with addiction may secretly consume rubbing alcohol as a substitute if they can’t get alcoholic beverages.
Encourage individuals struggling with addiction to seek professional help or rehabilitation.
Treatment of confirmed cases of poisoning usually involves supportive therapy. It may include fluid replacement therapy, oxygen supplementation, and vitamin supplementation in cases of suspected alcoholism-related malnutrition.
Treatment may also occasionally require hemodialysis to reduce the concentration of the toxic products in the victim’s body.
